14.6: Buffers - Chemistry LibreTexts
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the …
A solution containing a mixture of an acid and its conjugate base, or of a base and its conjugate acid, is called a buffer solution. Unlike in the case of an acid, base, or salt solution, the hydronium ion concentration of a buffer solution does not change greatly when a small amount of acid or base is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).

Buffers - Chemistry LibreTexts, PDF, Buffer Solution
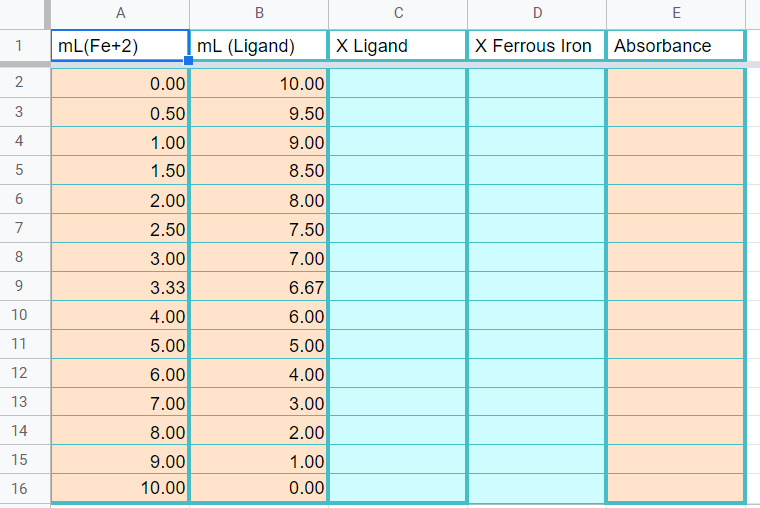
8.2: Formation Constants Lab - Chemistry LibreTexts
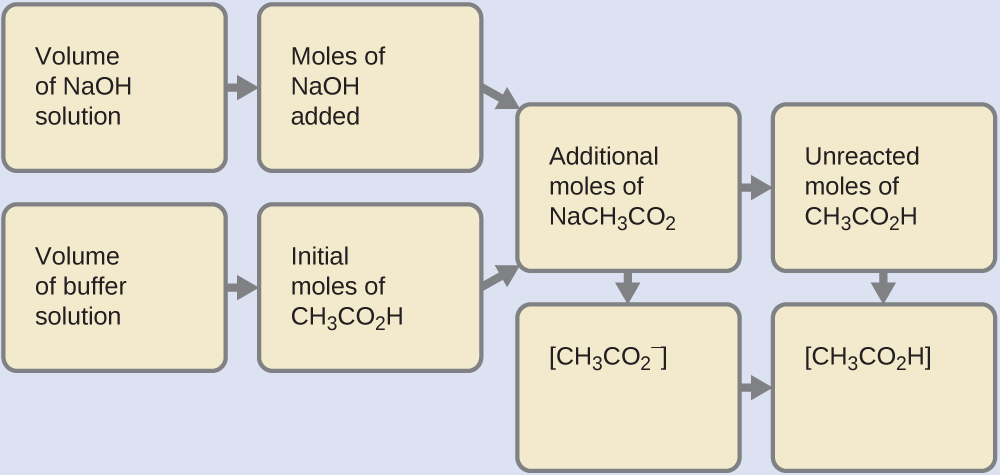
14.6 Buffers – Chemistry 112- Chapters 12-17 of OpenStax General

CHEM1106 - Experiment 1 Acids Bases And Buffers.pdf - Experiment 1
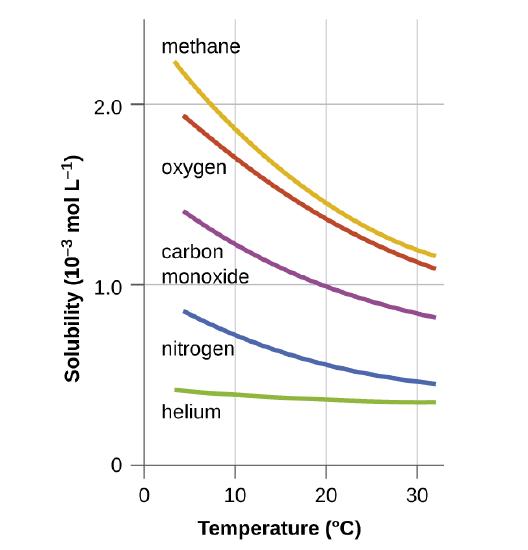
13.4: Solutions of Gases in Water- How Soda Pop Gets Its Fizz
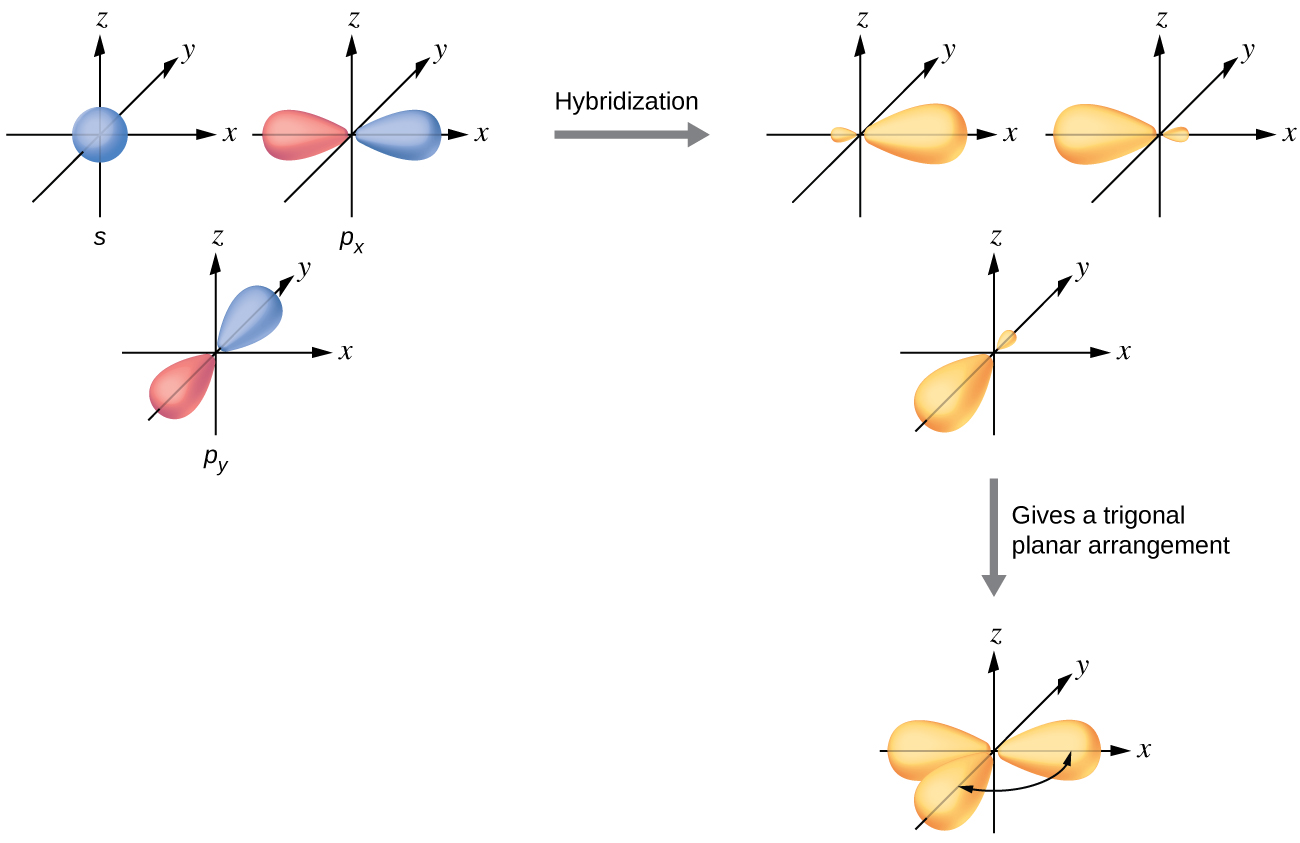
8.2 Hybrid Atomic Orbitals – Chemistry

5.1: Day 36- Buffer Solutions - Chemistry LibreTexts
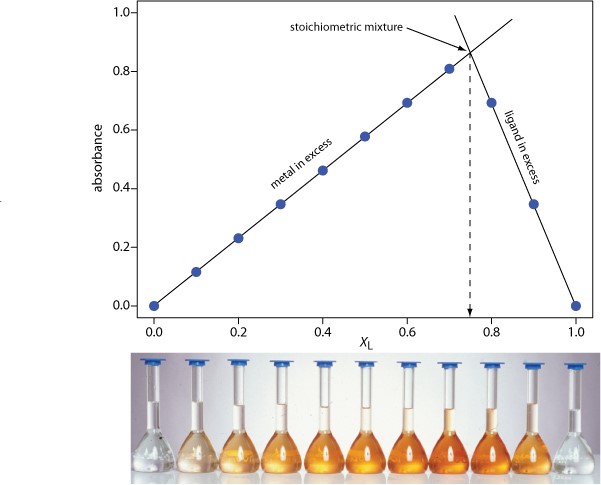
8.2: Formation Constants Lab - Chemistry LibreTexts

Chapter 16.6: Buffers - Chemistry LibreTexts