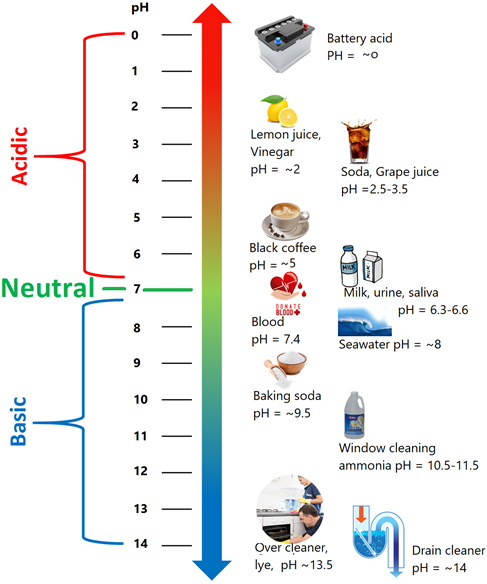
6.6: The pH - Chemistry LibreTexts
pH, i.e., log of reciprocal of hydronium ion concentration, its and measurement using pH paper and pH indicator is described. The importance of and pH of body fluid, acid rain, and its effects are …
pH, i.e., log of reciprocal of hydronium ion concentration, its and measurement using pH paper and pH indicator is described. The importance of and pH of body fluid, acid rain, and its effects are introduced.
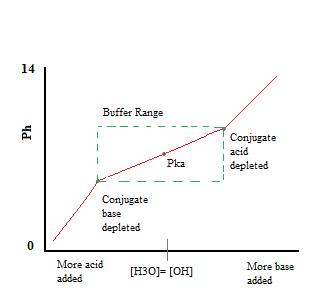
Introduction to Buffers - Chemistry LibreTexts

15.7: An Introduction to pH - Chemistry LibreTexts
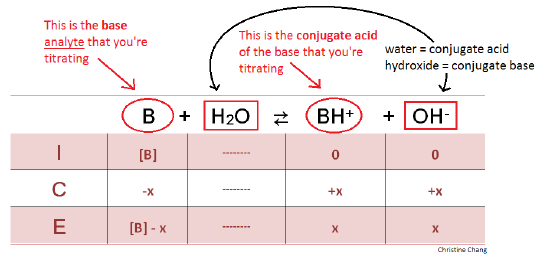
Titration of a Weak Base with a Strong Acid - Chemistry LibreTexts
18.1: Periodicity - Chemistry LibreTexts

pK-Yay: A Black-Box Method Using Density Functional Theory and Implicit Solvation Models to Compute Aqueous pKa Values of Weak and Strong Acids

15.7: An Introduction to pH - Chemistry LibreTexts
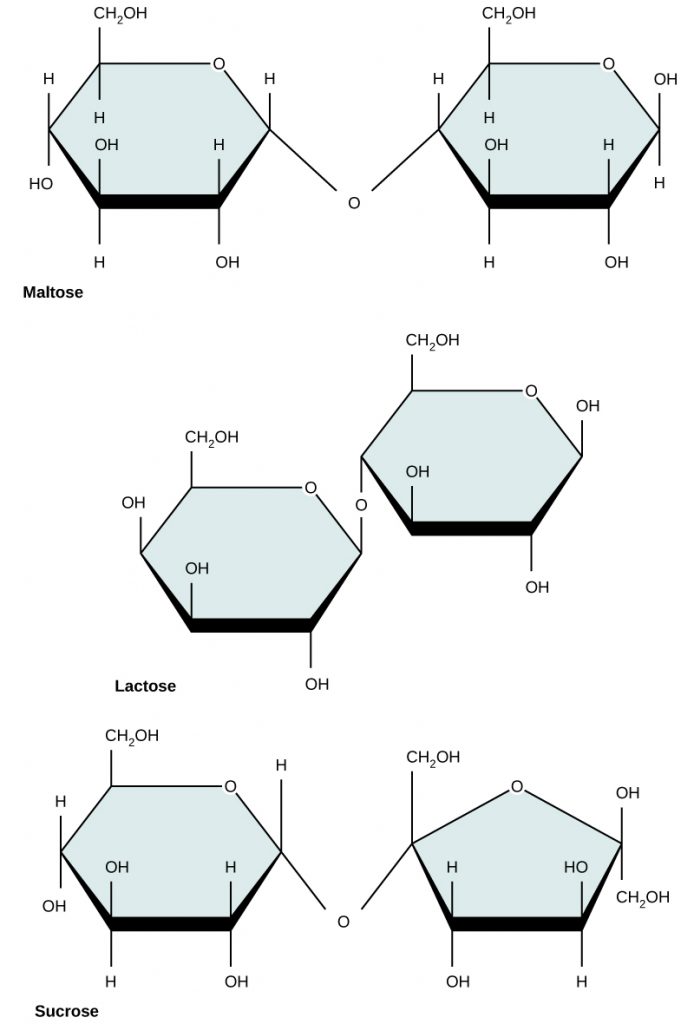
CH103 - Chapter 6: Natural Products and Organic Chemistry - Chemistry
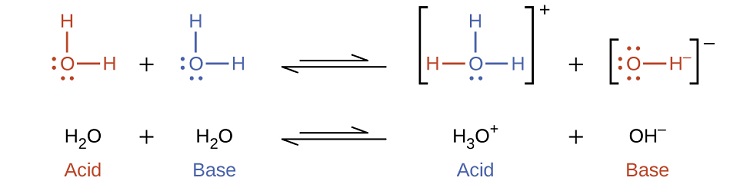
16.3: Self-Ionization of Water and the pH Scale - Chemistry LibreTexts
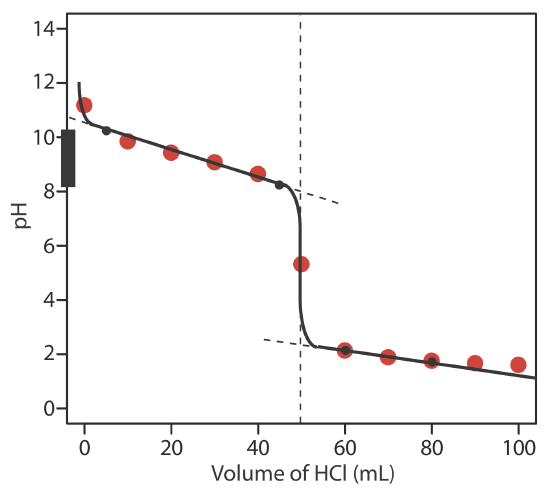
6.6: pH Calculations for Acid–Base Titrations - Chemistry LibreTexts

Heisenberg's Uncertainty Principle - Chemistry LibreTexts

3 Ways to Measure the pH of Water - wikiHow
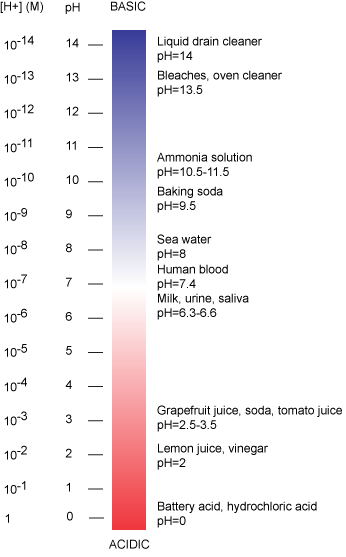
pH# - Biology LibreTexts

17.2: Buffer Solutions - Chemistry LibreTexts




/product/26/879143/2.jpg?3812)


