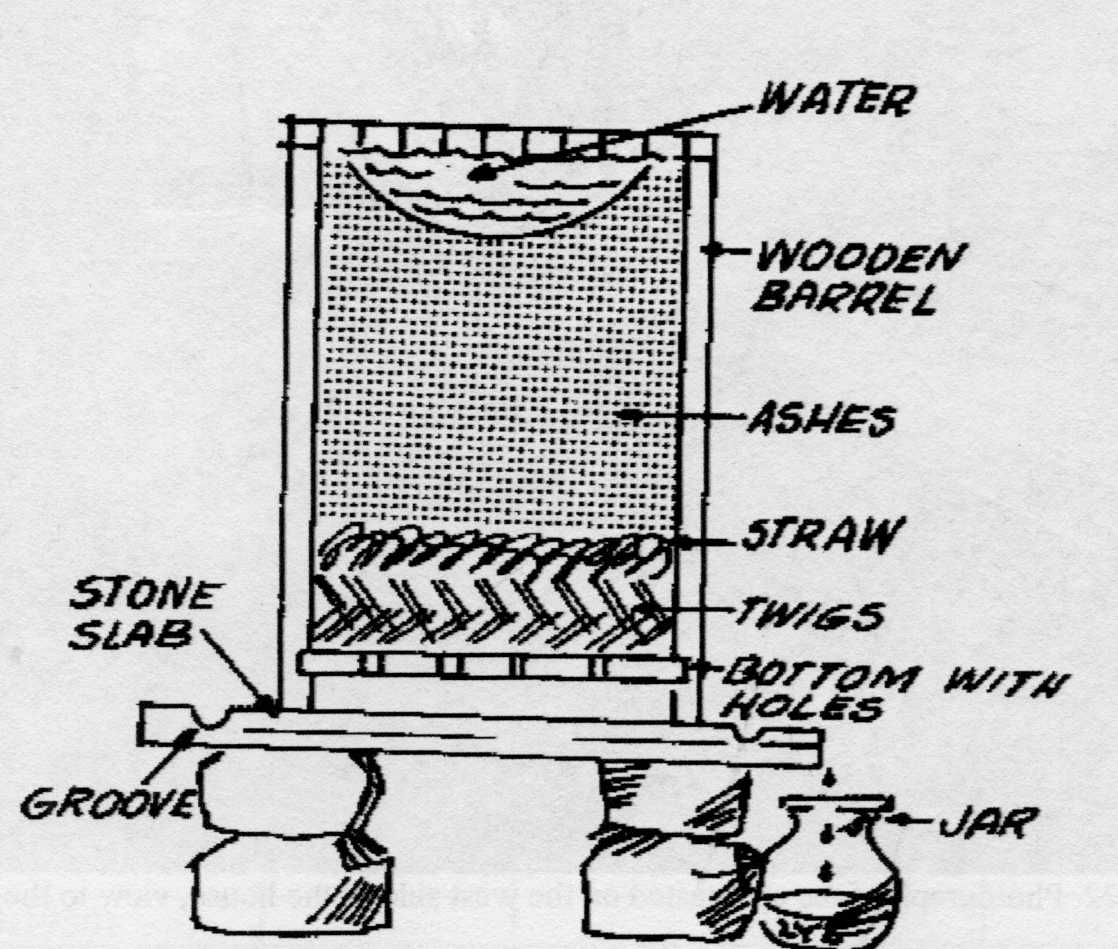
Boyer Recalls Six Brands of Sodium and Potassium Hydroxide Due to Failure to Meet Child-Resistant Packaging Requirement; Injuries Reported
Consumers should immediately store the recalled products in a safe location out of reach of children and contact Boyer for a free replacement child-resistant cap.

Powdered formula recalled due to possible bacteria contamination

marCH 2012 - Oser Communications Group

Full article: Abstracts of the European Association of Poisons Centres and Clinical Toxicologists XXIV International Congress

Toxicology Archives - REBEL EM - Emergency Medicine Blog

biOrigins Sodium Hydroxide Products Recalled Due to Failure to Meet Child-Resistant Packaging Requirement and Violation of FHSA Labeling Requirement; Imported by Madar Corporation; Sold Exclusively at .com (Recall Alert)

Navy Removal Scout 800 Pink Pill Assasin Expo Van Travel Bothell Punishment Shred Norelco District Ditch Required Anyhow, PDF

Midwest Lubricants Recalls Sodium Hydroxide Products Due to Failure to Meet Child-Resistant Packaging and FHSA Labeling Requirements (Recall Alert)

Full article: 2008 Annual Report of the American Association of Poison Control Centers' National Poison Data System (NPDS): 26th Annual Report
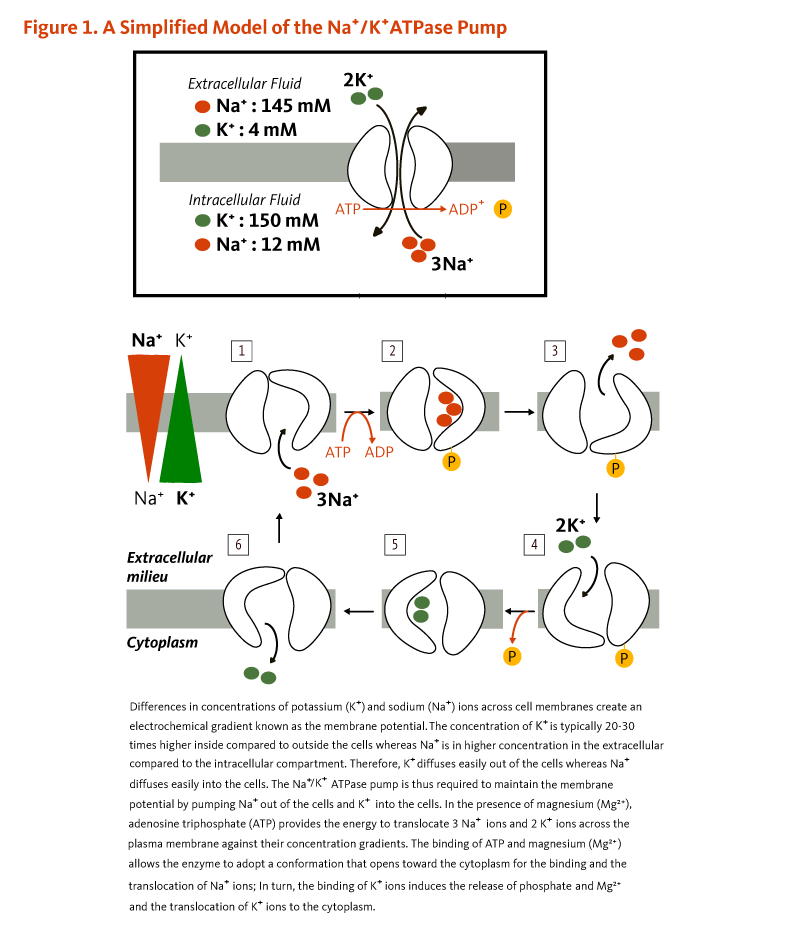
Minerals

PDF) 633 Field Performance of “Transgenic” Potato, with Resistance to Colorado Potato Beetle and Viruses

Children's Product Safety

Children's Product Safety

biOrigins Sodium Hydroxide Products Recalled Due to Failure to Meet Child-Resistant Packaging Requirement and Violation of FHSA Labeling Requirement; Imported by Madar Corporation; Sold Exclusively at .com (Recall Alert)