Difference Between Carbon and Graphite Compare the Difference Between Similar Terms
The key difference between carbon and graphite is that the carbon is a chemical element whereas the graphite is an allotrope of carbon. Carbon and graphite

Graphene & Graphite - How Do They Compare? – Graphenea
14.4A: Graphite and Diamond - Structure and Properties - Chemistry LibreTexts
What are the similarities and differences between graphite and

What is the Difference Between Alpha Carbon and Beta Carbon

What's the Difference Between Carbon Steel and Stainless Steel

How are Carbon Nanotubes Made from Graphene?
Q.Asseration:Diamond and graphite do not have the same crystal structure. Reason: Diamond is crystalline while graphite is amorphous

difference between diamonds and graphite

What is a Lithium-ion Battery? What are the Types of Lithium-ion Batteries?

What is the Difference Between Allotropes and Polymorphs
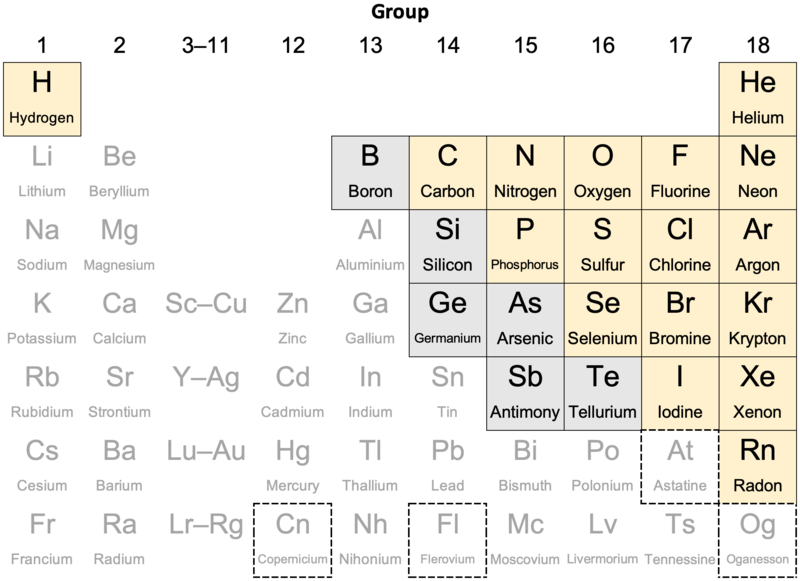
Nonmetal - Wikipedia

Explain the difference between diamond and graphite that what makes the graphite opaque to light while diamond is transparent to - Science - Carbon and its Compounds - 10691577

Difference Between Carbon and Graphite

Graphite Felt & Carbon Felt - CeraMaterials
Difference Between Silicon and Carbon
.jpg)