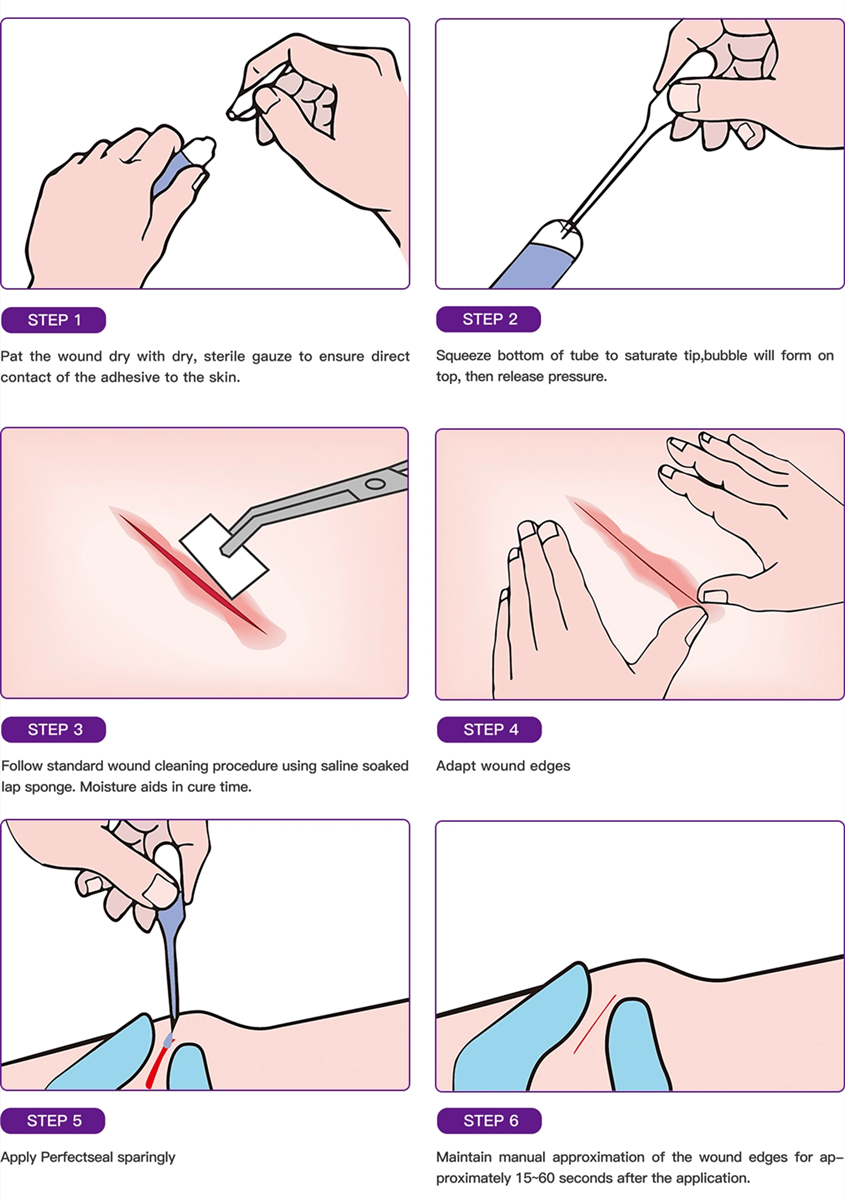
2-Octyl Cyanoacrylate (Dermabond) Wound Adhesives: Product, Design
The wound adhesive 2-octyl cyanoacrylate (Dermabond) is approved by the US Food and Drug Administration (FDA) for closure of incised skin. In addition to its surgical adhesive indication, 2-octyl cyanoacrylate was approved by the FDA in January 2001 for use as a barrier against common bacterial microbes, including certain staphylococci, pseud

Cyanoacrylate - Wikipedia
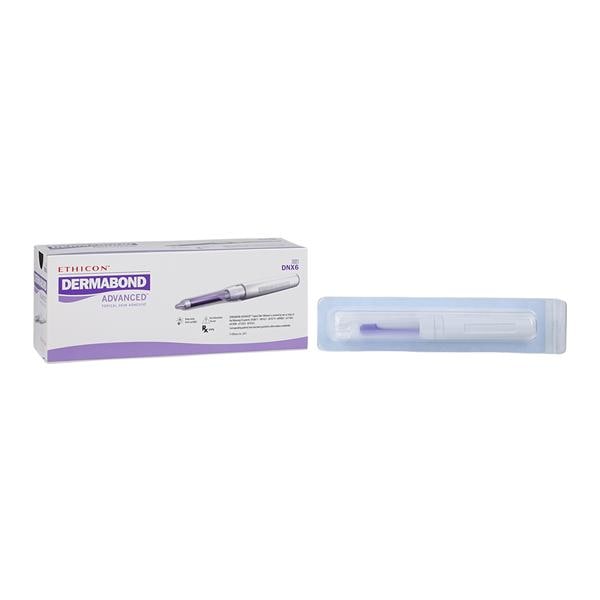
2-Octyl cyanoacrylate (Dermabond, Ethicon, Inc.)
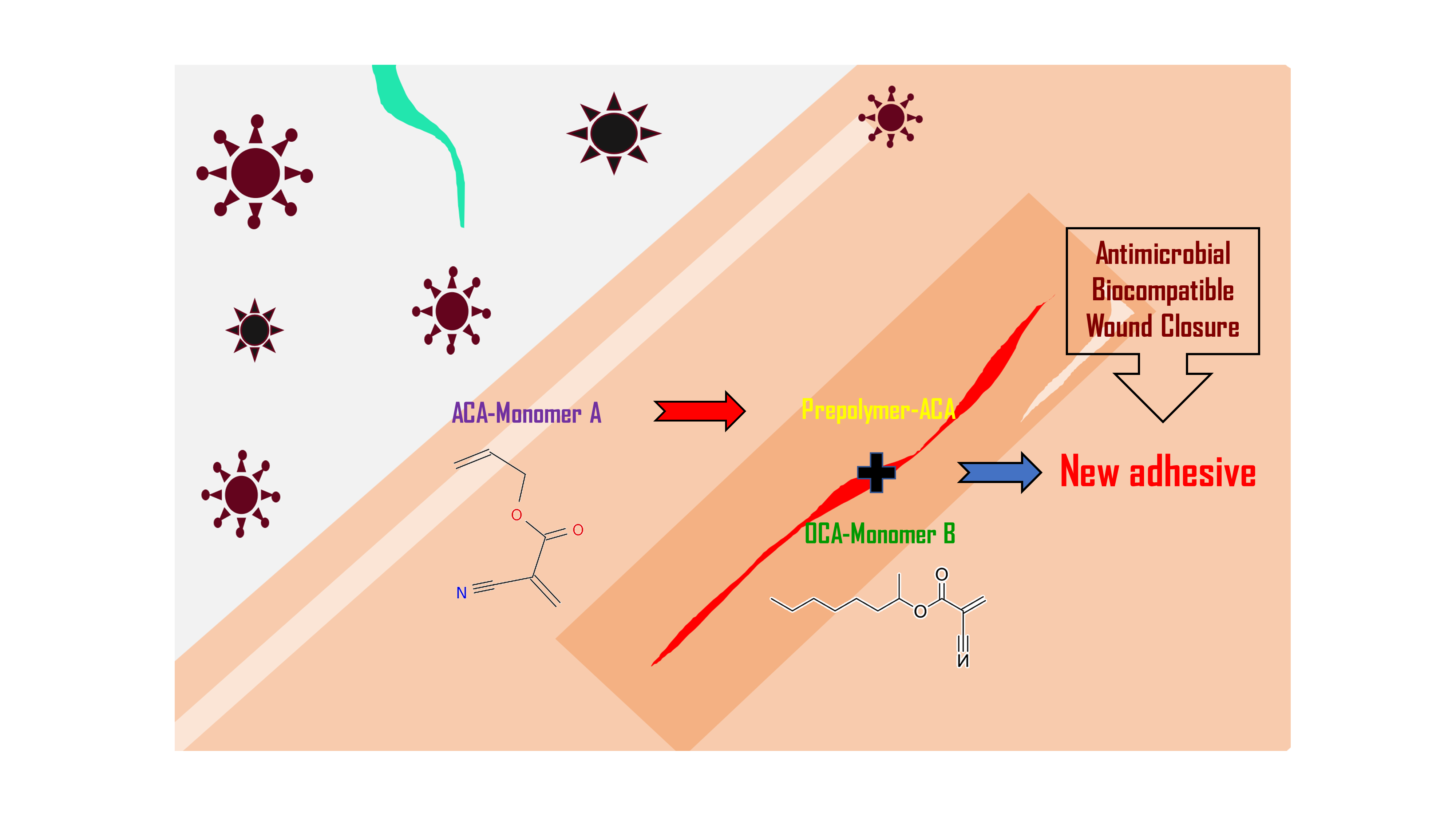
Materials, Free Full-Text

SKIN CLOSURE SYSTEM, PRINEO ADH (2/BX)
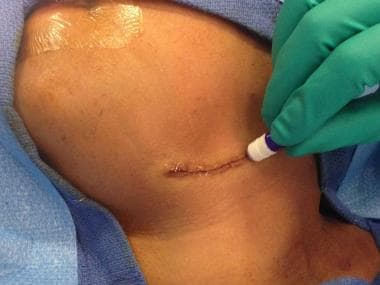
Precision Closure: Dermabond Mini 0.36 mL is designed for the precise closure of easily approximated, small incisions, and lacerations, offering a fast and efficient solution for wound closure. Inhibits Bacterial Growth: This skin adhesive demonstrates in vitro inhibition of both gram-positive bacteria (MRSA and MRSE) and gram-negative bacteria (E. coli), providing an added layer of protection against infection.

Dermabond Mini Topical Skin Adhesive
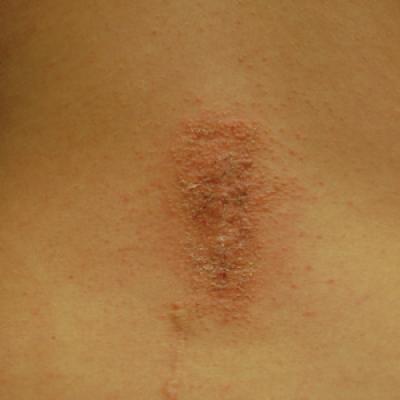
Allergic Contact Dermatitis to 2-Octyl Cyanoacrylate

Dermabond Advanced Directions for Use - Delasco

McKesson Brand 122-LBX - McKesson Medical-Surgical

Amtech Medical - Ethicon Dermabond Skin Adhesive