FDA Grants Import Discretion of Bracco's Iodinated Contrast Medium Iomeron (iomeprol injection) to Address Supply Shortages
Bracco Diagnostics Inc. has announced that the U.S. Food and Drug Administration (FDA) granted import discretion of Iomeron (iomeprol injection) into the U.S. to address the ongoing iodinated contrast media shortage. The product addresses the need for the most advanced diagnostic imaging standards and will be temporarily available in the U.S. market starting at the end of August, 2022.
Bracco Acquires Contrast Injector Maker Swiss Medical Care

Canon's Digital Upgrade Kit Enhances Efficiency
Contrast Media Imaging Technology News - 阿根廷vs乌拉圭直播
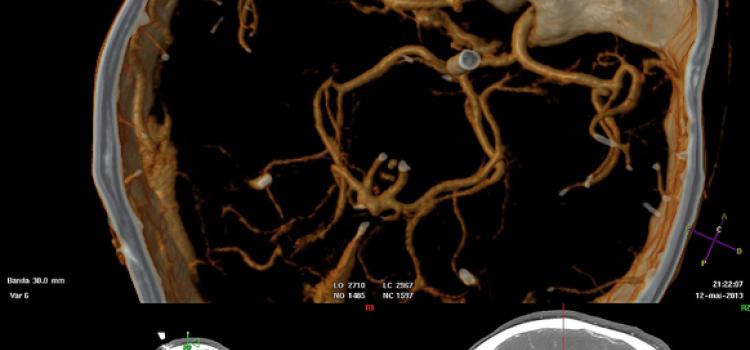
Volume Navigation 3-D Roadmap Aids More Accurate Interventions

Orthopedic Institute Selects Konica's Complete Digital X-ray Suite

Imaging Biometrics Submits FDA 510(k) Application for IB Zero G

FDA Grants Import Discretion of Bracco's Iodinated Contrast Medium Iomeron (iomeprol injection) to Address Supply Shortages
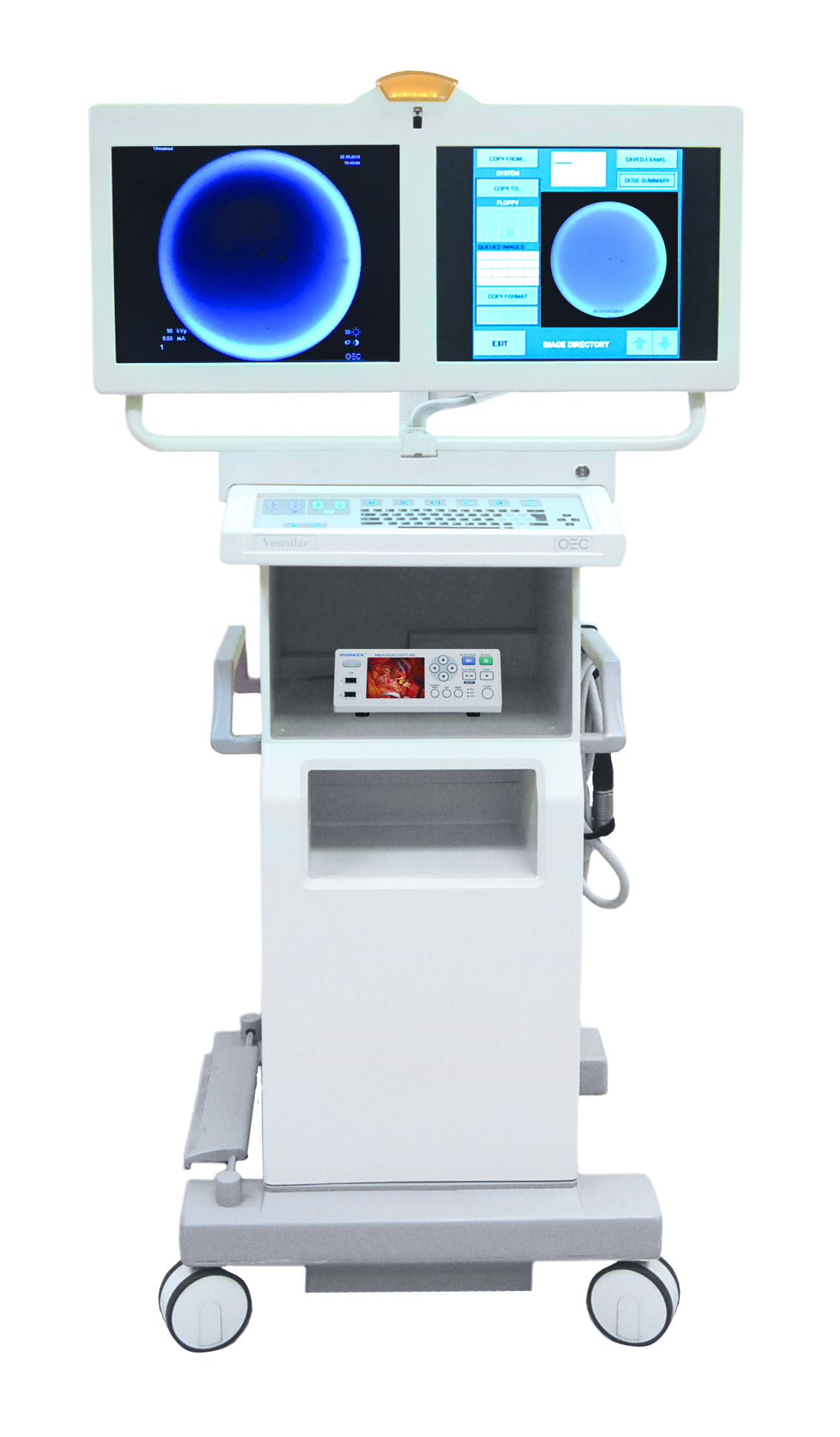
Ampronix Announces Availability of new OEC 9800 Retrofit Kit
Toshiba Highlights Advances in Angiography Imaging Systems

ACCU-PRO Offers Basic, Advanced Functions