Polysorbates 20 and 80 Used in the Formulation of Protein

Full article: A Platform analytical method for intact polysorbates in protein-containing biopharmaceutical products via HPLC-CAD
What's the Difference Between Tween 20 and Tween 80?

Factors affecting the physical stability (aggregation) of peptide therapeutics

Differences between polysorbate 20 and polysorbate 80 - Guangdong Huana Chemistry Co., Ltd.
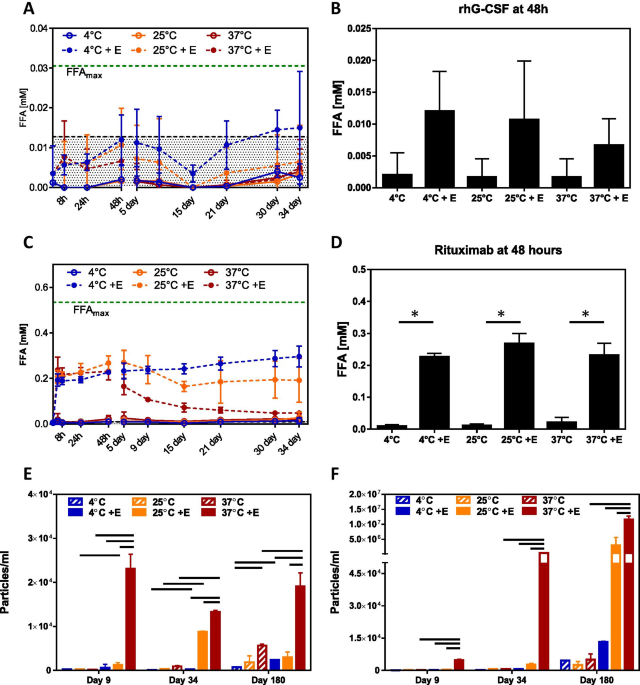
Effect of Fatty Acid Composition in Polysorbate 80 on the Stability of Therapeutic Protein Formulations
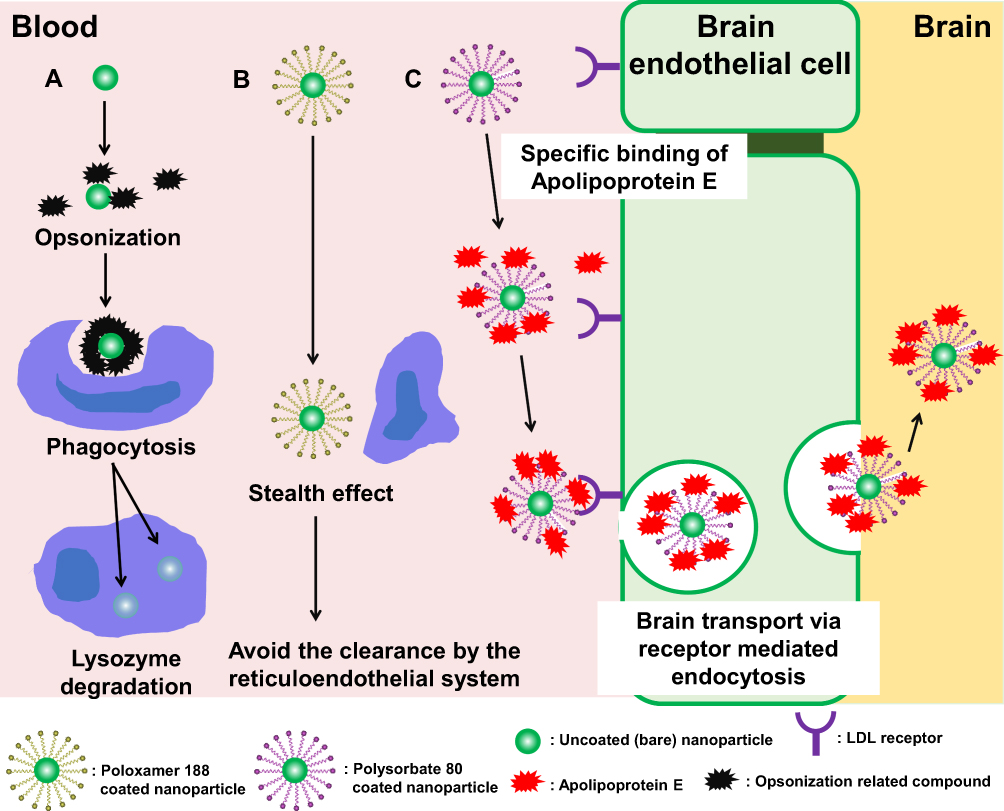
Surfactant-coated Nanoparticles in Nanomedicine and Food

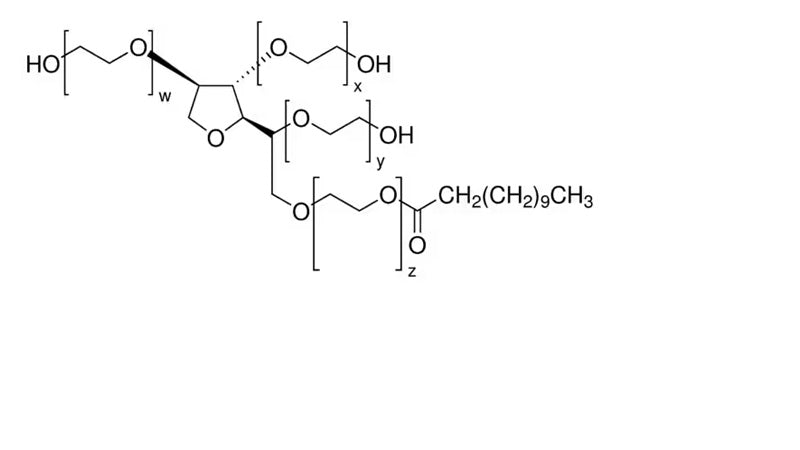
Figure 3 from Polysorbates 20 and 80 used in the formulation of protein biotherapeutics: structure and degradation pathways.

Quantitative Analysis of Polysorbate 20/80 in Protein-Based Biopharmaceuticals Using A One-Pot RPLC-MS Based Platform Method

Quantifying, Profiling Polysorbates with Charged Aerosol Detection - Analytical Methods
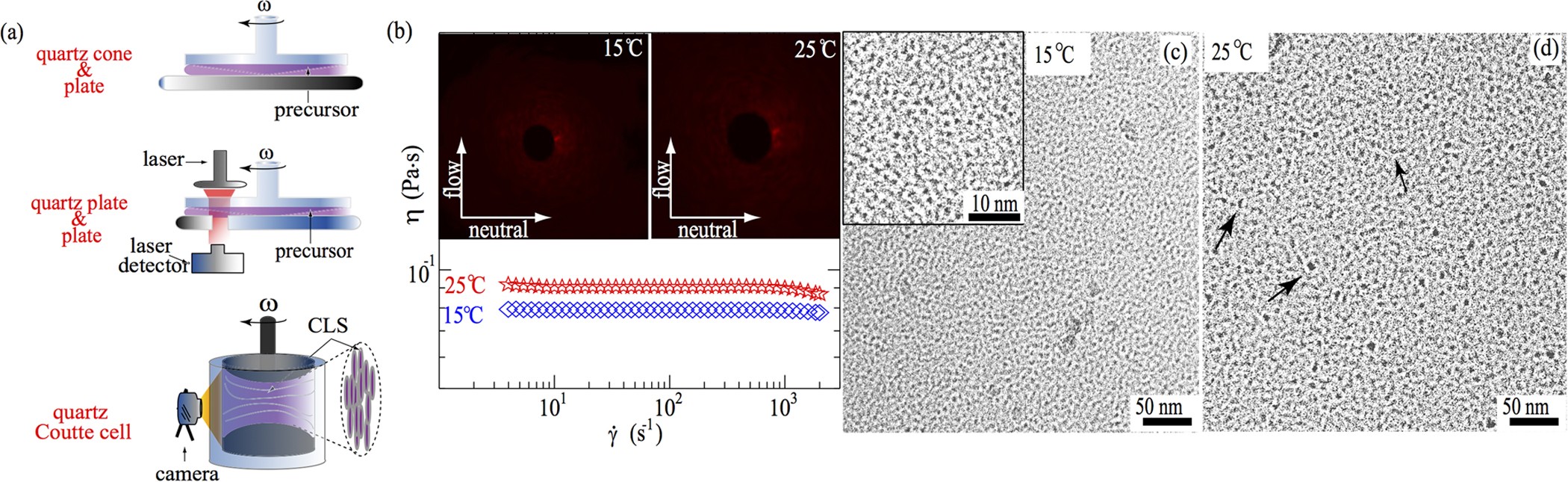
Formation of crystal-like structures and branched networks from nonionic spherical micelles
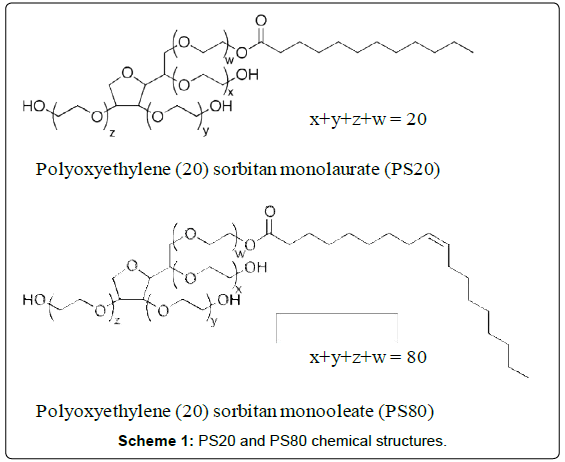
A Highly Sensitive Method for the Quantitation of Polysorbate 20 and 80 to Study the Compatibility between Polysorbates and m-Cresol in the Peptide Formulation

The degradation of poloxamer 188 in buffered formulation conditions, AAPS Open

Synthesis and Characterization of Lyophilized Chitosan-Based Hydrogels Cross-Linked with Benzaldehyde for Controlled Drug Release

Applied Sciences, Free Full-Text