Buffer Solutions: Definition, Types, Preparation, Examples and Videos
Buffer Solutions: What do you think will happen if the pH of our blood changes drastically from its normal pH of 7.35? Yes, the cells of our body will not function properly and our body systems will fail! Human blood contains a 'buffer' that allows it to maintain its pH at 7.35 to ensure normal functioning of cells.

Buffer Solutions
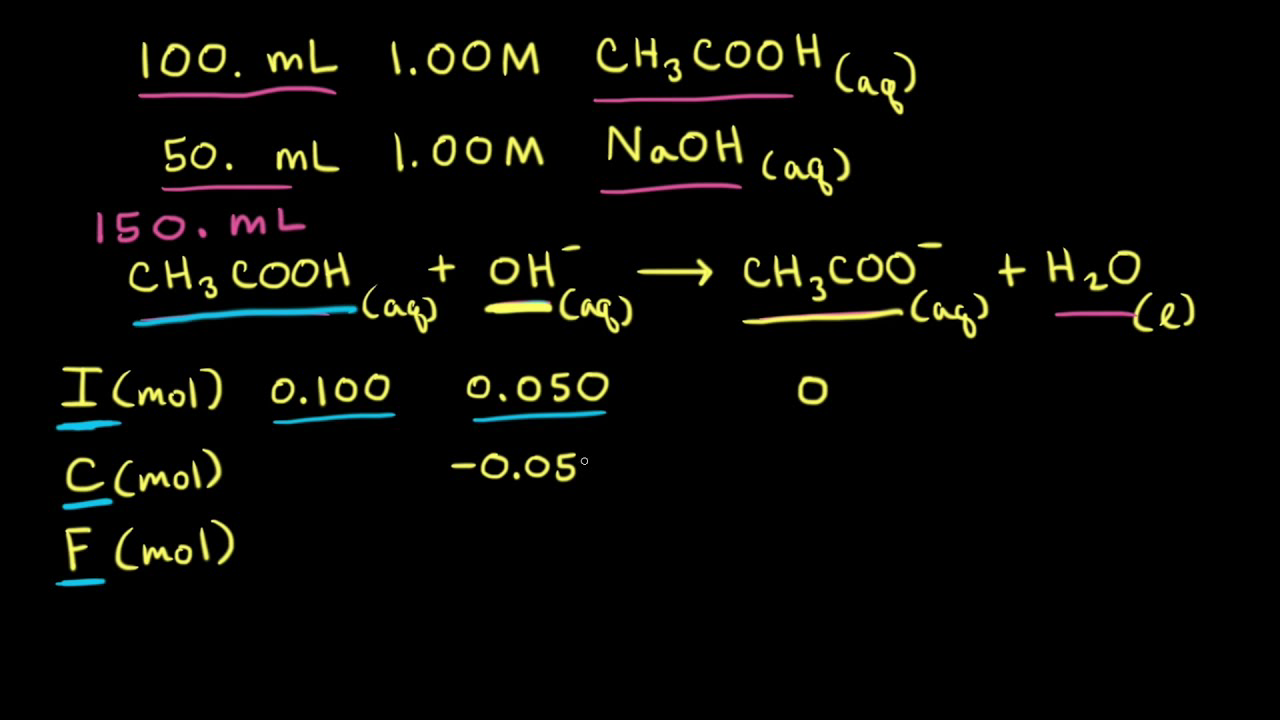
Methods for preparing buffers (video)

Calculate pH of Buffer Solution

Types of Employee Benefits: 12 Benefits HR Should Know - AIHR

Buffer Solutions: Preparation, Mechanism, Types, and Uses
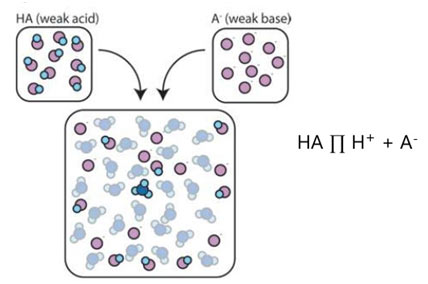
Buffer Solutions - Definition, Types, Working, Preparation & Significance

Z-buffering - Wikipedia

Chemistry!!! Not Mystery : How to Prepare Buffer Solutions?

Guide to the Buffer Strategy

What if Buffer Solution, Types of Buffer Solution

An acidic buffer solution can be prepared by mixing solution of

Buffer System in Chemistry, Definition, Function & Examples - Video & Lesson Transcript
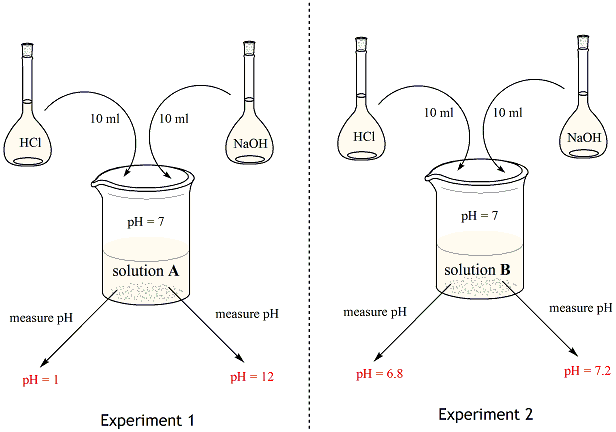
Chemistry of buffers and buffers in our blood (article)
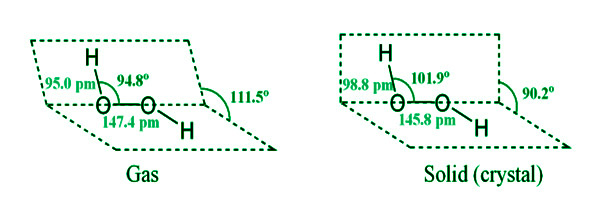
Hydrogen Peroxide - Uses, Properties, Preparation, Examples - GeeksforGeeks