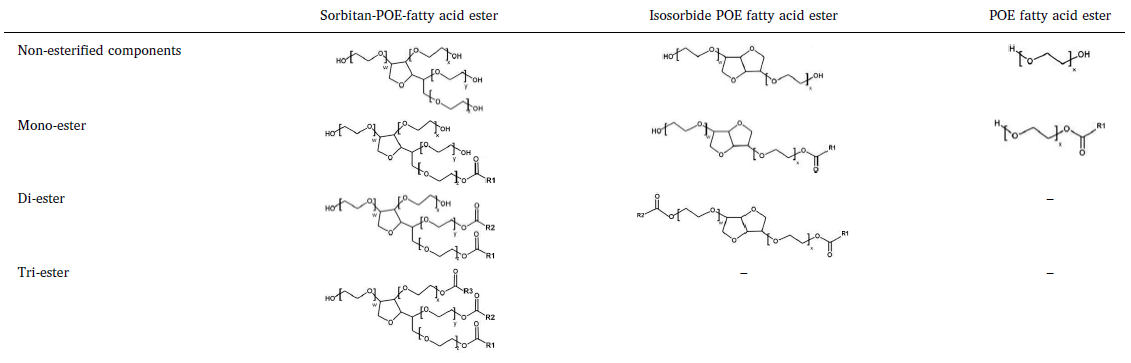
Development and validation of a selective marker-based quantification of polysorbate 20 in biopharmaceutical formulations using UPLC QDa detection - Pharma Excipients
Polysorbates are widely used as non-ionic surfactant in biopharmaceutical formulations. Recently, the degradation of polysorbate moved into the focus of
Polysorbates are widely used as non-ionic surfactant in biopharmaceutical formulations. Recently, the degradation of polysorbate moved into the focus of attention, because in several published studies it was described, that stability issues in polyso

Polysorbate 20 Degradation in Biopharmaceutical Formulations: Quantification of Free Fatty Acids, Characterization of Particulates, and Insights into the Degradation Mechanism

Pharmaceutics, Free Full-Text

Quantitative Analysis of Polysorbate 20/80 in Protein-Based Biopharmaceuticals Using A One-Pot RPLC-MS Based Platform Method

Hydrolytic polysorbate 20 degradation – Sensitive detection of free fatty acids in biopharmaceuticals via UPLC-QDa analytics with isolator column - ScienceDirect

Pharmaceutics, Free Full-Text

Development of a High-Throughput Formulation Screening Platform for Monoclonal AntibodiesBioProcess International
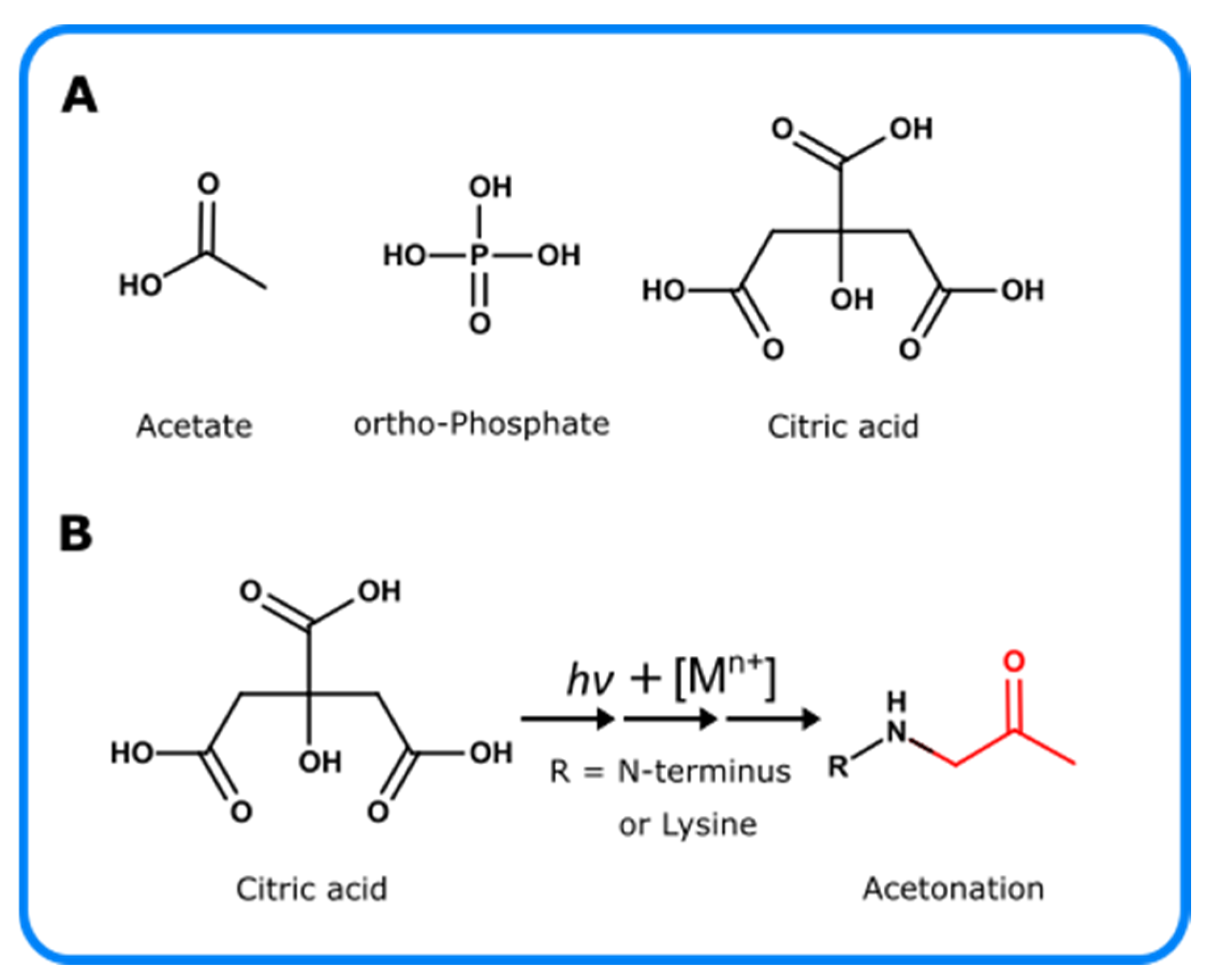
Pharmaceutics, Free Full-Text

A Platform analytical method for intact polysorbates in protein-containing biopharmaceutical products via HPLC-CAD: Journal of Liquid Chromatography & Related Technologies: Vol 45, No 17-20
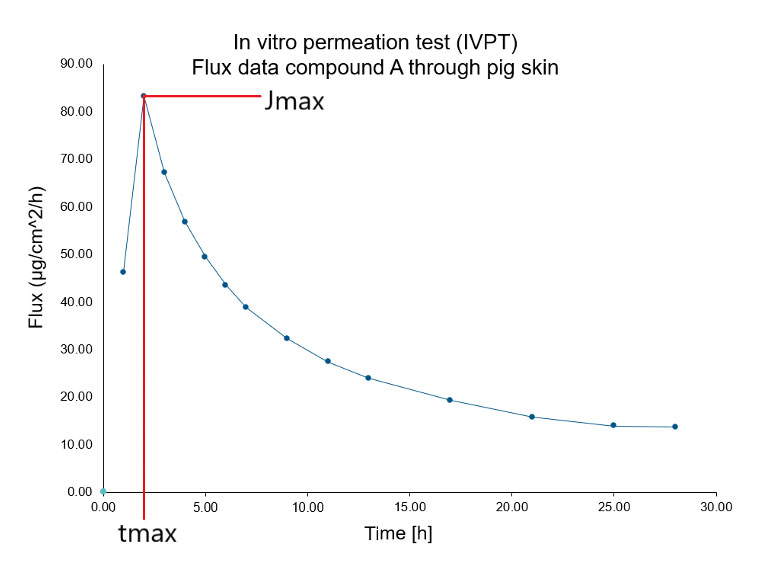
RaDes EN Archive - RaDes GmbH

Characterization and Stability Study of Polysorbate 20 in Therapeutic Monoclonal Antibody Formulation by Multidimensional Ultrahigh-Performance Liquid Chromatography–Charged Aerosol Detection–Mass Spectrometry
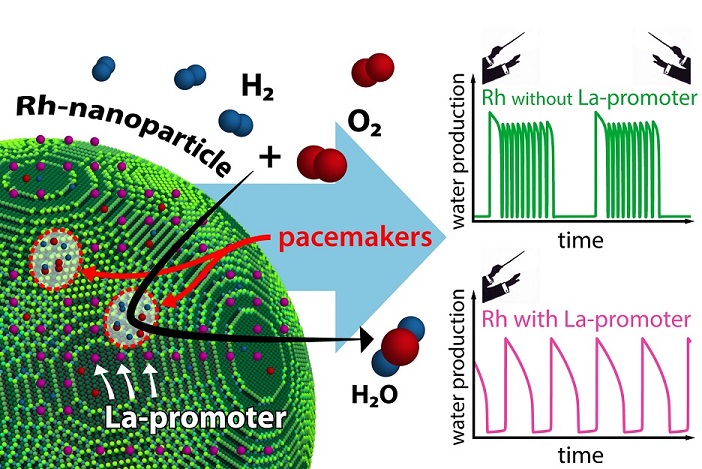
promotercatalystimage-1700816724160.jpg

Photo-Oxidation of Therapeutic Protein Formulations: From Radical Formation to Analytical Techniques. - Abstract - Europe PMC









